Half of over 50s, two-thirds over 70 have now had first COVID jab
A review of all aspects of the COVID-19 vaccine rollout plan will be completed within weeks, as an inquiry heard “hardly anyone” has received a second dose.
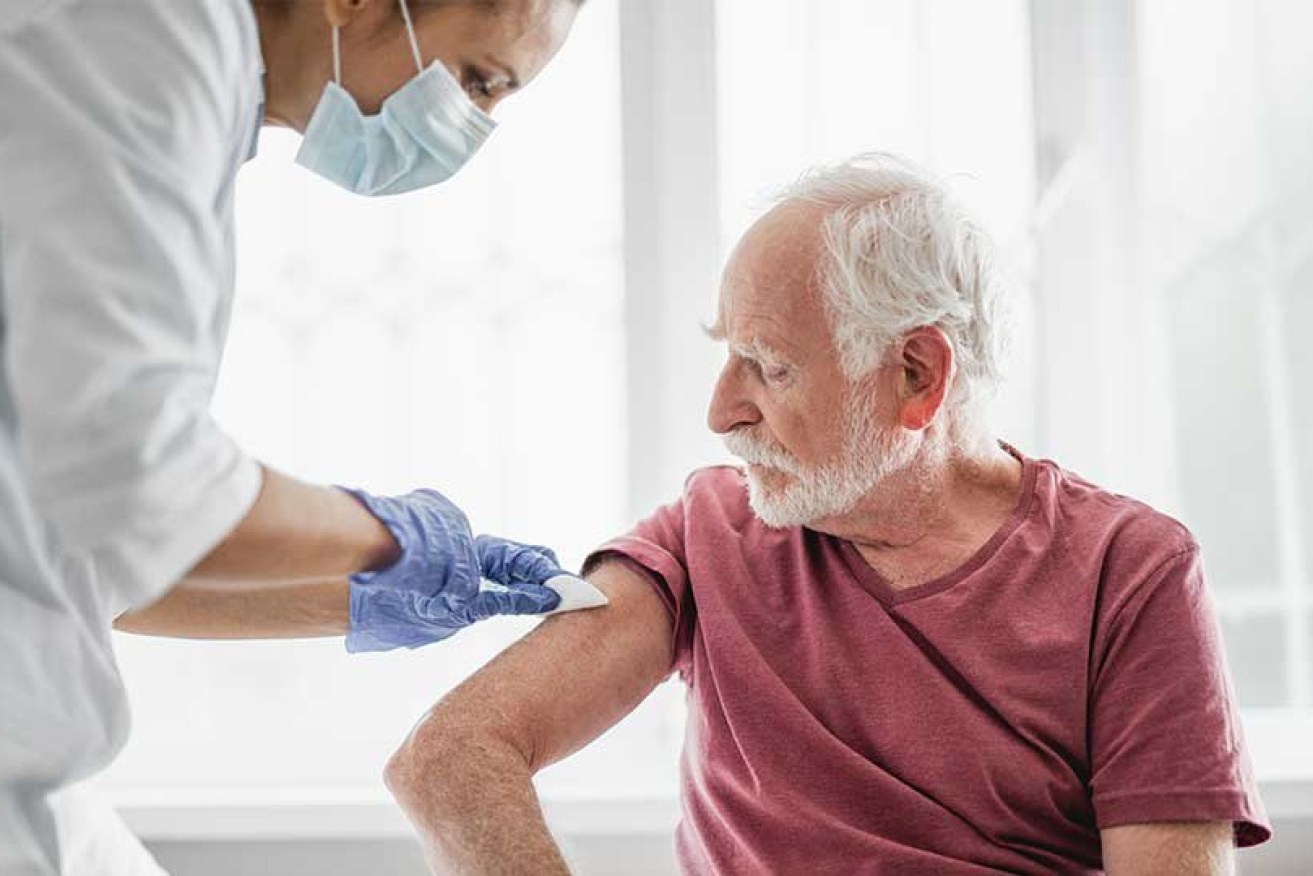
Mater researchers have found a longer protecting flu vaccine (Pic: supplied)
Lieutenant General John Frewen, the coordinator-general of the national COVID vaccine task force, told a Senate committee looking at the pandemic response he had been brought in to test “all planning assumptions”.
“We are now looking at all aspects of the rollout plan and looking for any opportunities we will have to accelerate the rollout through the year as supplies come on line,” he said.
The review would also include the use of pharmacists to provide vaccinations.
Health department secretary Brendan Murphy said a “complete overview” of the plan was needed to ensure the substantial ramp-up in the second half of the year went smoothly.
Murphy told the committee 48 per cent of over-50s had received a first jab, while 65 per cent of over 70s had their initial shot.
However, Professor Murphy added: “Hardly anyone has had their second shot because the AstraZeneca has a 12-week schedule.”
“It only started second shots in the last two weeks,” he said.
He said it was too early to say whether the new advice on AstraZeneca being limited to over-60s would lead to hesitancy, but the message remained clear that the risk of a blood clot following a second dose was “miniscule”.
“We haven’t seen a discernible drop-off,” he said.
General Frewen said he did not expect a major disruption from the adjustment as the new advice about AstraZeneca kicks in and Pfizer is more broadly rolled out.
He said there were 2.3 million doses of Pfizer on hand and it was expected another 3.4 million would be available over the next six weeks.
Trials are under way in the UK, Spain, Germany and the US on whether it is safe and effective to allow people to get a different type of vaccine – so-called “mixing and matching” – when it comes to their second dose.
Therapeutic Goods Administration chief Professor John Skerritt said it could be two months before the results of the trials are available, after which he expected submissions would be made to the TGA.
“What is known is with a combination of the two you do get a better antibody response and your cellular immune system seems to be quite well primed, but that is far short of showing there is good protection against hospitalisation, death or … catching the disease.”
Professor Murphy said in the meantime people should get their second dose of what they intially received.
Professor Skerritt said he expected to receive an application from Moderna for another vaccine for use in Australia in the first week of July.
It was likely the advice could cover its use for teenagers.
Novavax data was not expected until September.












