Local bio-tech firms break into new COVID markets
Two Brisbane bio-techs have broken through to win new markets for their COVID-19 test kits, including the current global hotspot of Indonesia.

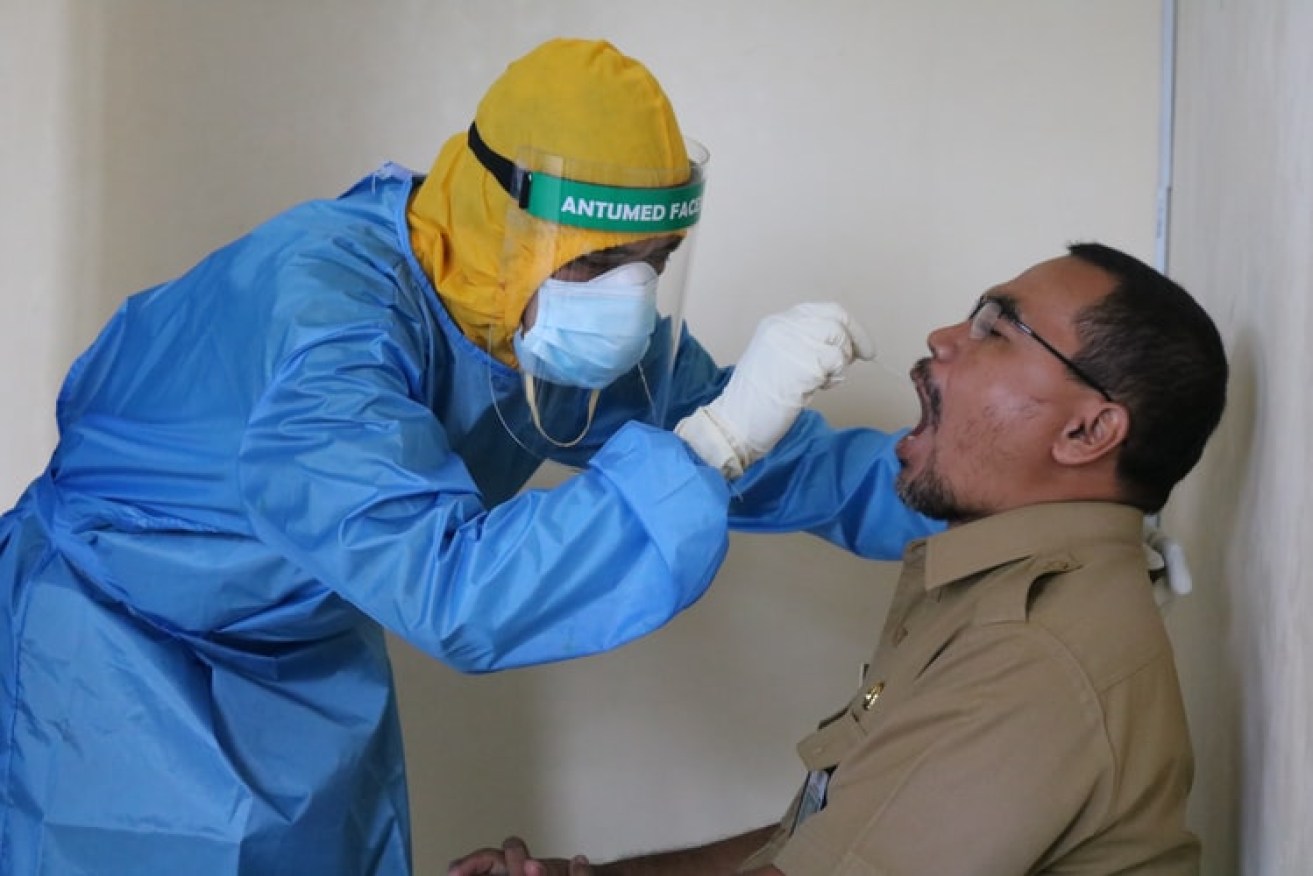
Two Brisbane companies have broken into new markets for their COVID test kits.
AnteoTech has struck a deal with BioMed Global for exclusive sales into Malaysia, Singapore, Indonesia, Vietnam, Thailand and Myanmar.
It coincided with Ellume announcing it had received approval from the US Food and Drug Administration to sell to point of care providers and mass testing sites. It previously had approval to sell over the counter.
It has also been selling to airlines which then allow passengers to use the technology to provide a COVID-free status for entry into the US.
Anteotech chief executive Derek Thomson said the Biomed deal was a significant milestone for the rollout of its mass testing EuGeni platform.
“This agreement will provide is access to six major south-east Asian territories covering a total population of around 340 million people,” Thomson said.
He said Biomed would be selling into laboratories, hospitals and major testing centres.
“Asian borders are starting to open up and this process requires rapid testing to ensure transiting people are not transmitting COVID-19. We will work with Biomed closely to offer EuGeni as a COVID-19 screening tool to ensure we maximise the potential of these opportunities,” Thomson said.
Anteotech had sufficient manufacturing capacity to meet immediate demand and any increase could be dealt with through its current expansion of capacity.
It also announced a distribution agreement for New Zealand through Abacus Pty Ltd and is also going through its compliance audit and other accreditation to allow for a submission to the Therapeutic Goods Administration for the EuGeni platform in Australia.
Its submission is due in September.
“We have already received strong interest from the private sector for COVID-19 screening opportunities domestically and in the Pacific and we will be working with Abacus over the coming months to further promote EuGeni in those settings,” Thomson said.
He said the market in Australia and New Zealand were small but important. However, the bigger opportunities were in Asia and Europe.
AnteoTech also sells its AnteoBind technology to Ellume for use in its test kits.












